
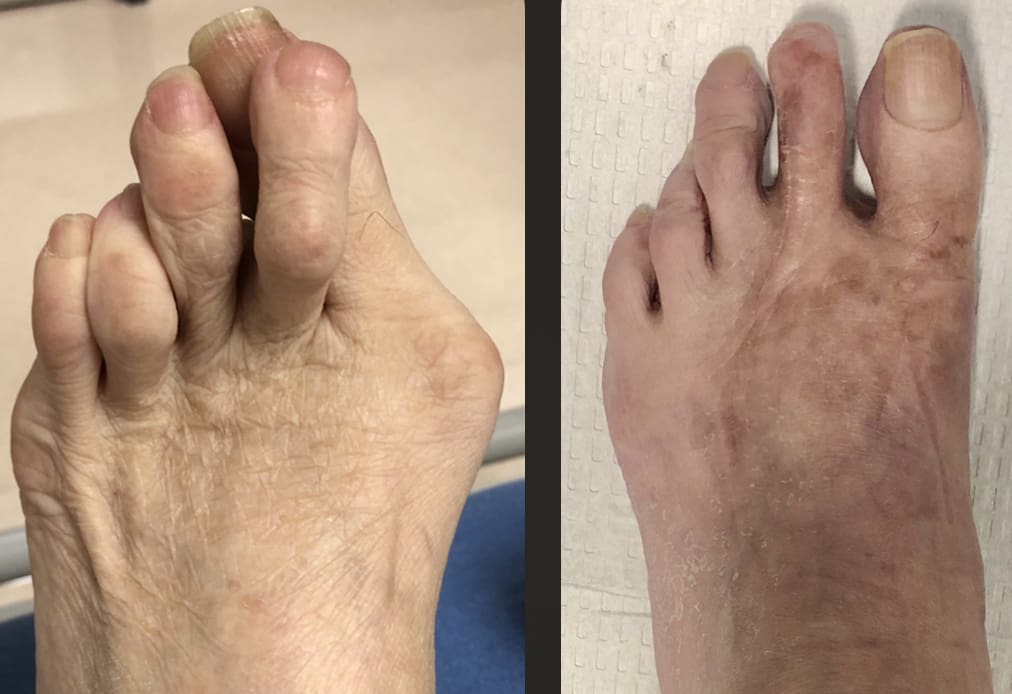
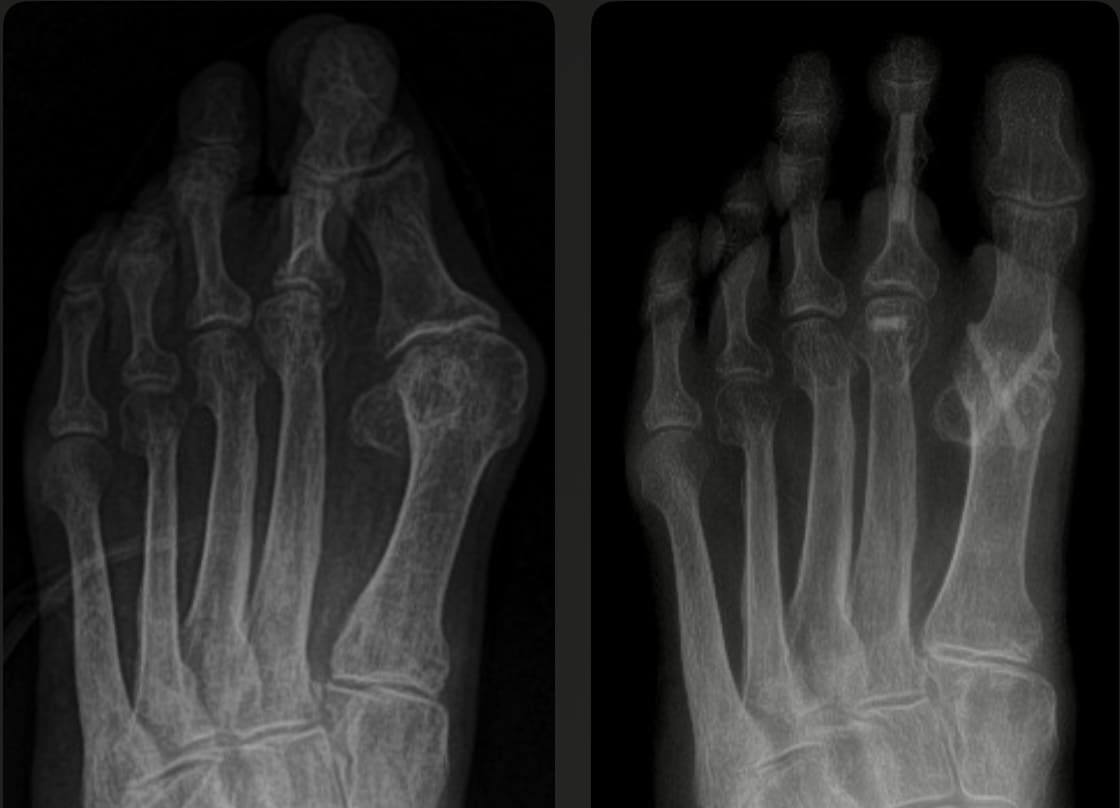
The Bone Pin...
One solid piece of 100% human cortical bone (allograft) processed in a dowel configuration - no bio-engineering necessary to the AlloMate Bone Pin due to the appropriate hole diameter and the “pound-in type” insertion technique achieved by the AlloMate Bone Pin System.
Integrates via a well-known process of "creeping substitution."
Can be easily identified on x-ray studies.
Alternative to metal, PEEK, and resorbable devices.
Rationale of the AlloMate Bone Pin System:
"The most logical way to closely mimic the physiological bone healing process while also stabilizing the osseous construct in a safe, reproducible and cost-effective manner."
Human allograft cortical bone pins allow surgeons to address
a wide variety of surgical applications.
- Resorbable: Eliminates the need for a second surgery to remove hardware.
- Patient Specific: AlloMate Bone Pins can be trimmed to size
to meet each patient’s specific need. - Single-Use: AlloMate procedure kits feature single-use
instruments that are always sterile and
ready for use at a moment’s notice.

Applications
AlloMate Bone Pins are suitable for a range of orthopedic and trauma applications, including, but not limited to:
Osteochondral Dissecans Lesions
Knee, Ankle, Elbow
Arthrodesis
Small joint, midfoot, hand
Other Applications
Hammertoe repairs, bunionectomies, osteotomies, Freiberg's infraction, flatfoot correction, non-union revision and backfill after hardware removal
Fracture Repair
Metatarsal, phalangeal, midfoot, avulsion, medial malleolar, calcaneal, stress reaction, distal radius


SINGLE-USE. STERILE PACKED. READY TO USE
COST SAVINGS
• Eliminates the need for metal trays, thereby
eliminating washing, sterilization and
decontamination time and cost.
• The average cost to process reusable instruments
for a single case is approximately $285. (1)
REDUCED INFECTION RISK
• Potential reduction in non-sterile instrument
occurrence as all instruments within the AlloMate
System are sterile packed and single-use. (2)
• Surgical site infection is estimated at 21.8% of all
health care associated infections, costing on average
$16,000 - $21,000 per case. (1)
IMPROVED EFFICIENCY
• Streamlined component list. Only 13%-22% of
instruments within traditional metal reusable trays
are estimated to be used during the entirety of a
procedure. (1)
• Brand new, sharp, pristine instruments for every case.
• Potential reduction in surgical delays or cancellations
due to non-sterile, missing, or dysfunctional
instruments.
• Operating room costs are $36-$37 per minute, delays
related to missing or faulty instrumentation may
increase costs by approximately $260 per case.
[1] Apurva Shah, MD, MBA. The Value Proposition of Single-Use Sterile Procedure Kits. May 2021
[2] Siegel GW, Patel NN, Milshteyn MA, Buzas D, Lombardo DJ, Morawa LG. Cost Analysis and Surgical
Site Infection Rates in Total Knee Arthroplasty Comparing Traditional vs. Single-Use Instrumentation. The
Journal of Arthroplasty, Volume30, Issue 12, P2271-2274, December 01, 2015
